Electrochemical Characteristics Of Selected Molecules
- WHAT ARE CYCLIC VOLTAMMETRY DATA CHARTS?
- NOMENCLATURE
- CONDITIONS
- ELECTROCHEMICAL CHARACTERISTICS OF SELECTED MOLECULES
- AMINOPHENOLS
- AROMATIC AMINES
- ALKYLPHENOLS OF ENVIRONMENTAL INTEREST
- CHLOROPHENOLS OF ENVIRONMENTAL INTEREST
- VANILLYL METABOLITES OF TYROSINE AND RELATED COMPOUNDS
- INDOLE METABOLITES OF TRYPTOPHAN AND RELATED COMPOUNDS
- CATECHOLAMINES AND OTHER EASILY OXIDIZED DIPHENOLS
- PHENOLIC ACIDS
- SELECTED COMPOUNDS OF ENVIRONMENTAL INTEREST
WHAT ARE CYCLIC VOLTAMMETRY DATA CHARTS?
The following tables will help to summarize a great deal of information about the redox characteristics of a variety of molecules. This information can be used as a guideline when evaluating the feasibility of detecting these compounds using LCEC techniques. Information concerning the applied potential is not absolute; you will need to confirm the optimum using hydrodynamic voltammograms and the electrode and mobile phase necessary for your assay.
NOMENCLATURE
The potential axis runs from left to right for reductions and from right to left for oxidations, per the traditional American convention.
For REDUCTIONS: the left end of the rectangle represents the potential where the current is equal to 1/2 of the peak current. The right end of the rectangle represents the potential at the peak maximum. The opposite is true for OXIDATIONS.

An empty rectangle indicates that only one forward peak is observed and no other peak is obtained on the reverse scan between limiting potentials of the medium.

A shaded rectangle indicates that there is one reduction (or oxidation) peak and that an oxidation (or reduction) peak is observed when the scan is reversed. However, the electrochemical reaction is CHEMICALLY IRREVERSIBLE.
For example,

A solid rectangle means that the system is CHEMICALLY REVERSIBLE (and that the electron transfer system is sufficiently fast that both forward and reverse processes occur on the time scale of the CV experiment).

A rectangle without the right (or left for an oxidation) bar indicates that a poorly defined forward peak was obtained (a shoulder on the second wave) and that the E1/2 value was estimated by setting E1/2 at the potential equal to 1/2 of the current at the peak shoulder.


A solid bar following a rectangle indicates the peak potential of additional forward peaks that exist before the background limit. However, the E1/2 value cannot be determined due to the poor resolution between adjacent peaks.

For differential pulse and other small-amplitude voltammetric techniques, this symbol is used. The central vertical bar represents the peak potential and the box indicates the width of the curve at half the peak current. The same symbol is used for polarography and hydrodynamic voltammetry where the three vertical bars indicate the E1/4, E1/2 and E3/4 potentials. Again, a narrow box suggests a fast heterogeneous electron-transfer rate. An asymmetric box may indicate complications in the mechanism.
CONDITIONS
All cyclic voltammograms used to obtain data on the following 9 figures used identical conditions:
| Electrolyte: | 0.1 M Citrate:10% Ethanol (V:V) |
| Scan: | 200 mV/sec |
| Working Electrode: | Carbon Paste type CPO (for oxidative CV) |
| Mercury/Gold Amalgam (for reductive CV) | |
| Conditions: | Deoxygenation of solution was required for Reductive CV |
Figure 8.1. Oxidative Cyclic Voltammetric Data for Selected Aminophenols.
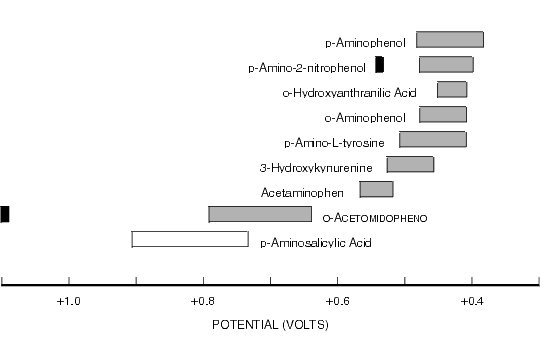
Figure 8.2. Oxidative Cyclic Voltammetric Data for Selected Aromatic Amines.
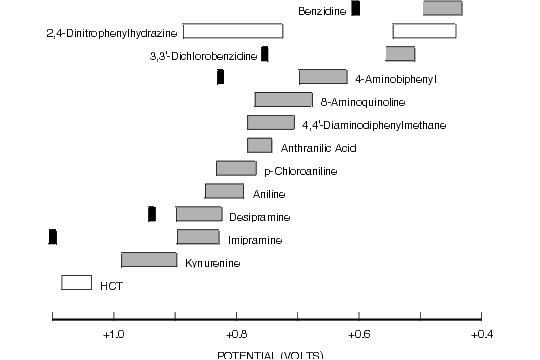
Figure 8.3. Oxidative Cyclic Voltammetric Data for Selected Alkylphenols of Environmental Interest.
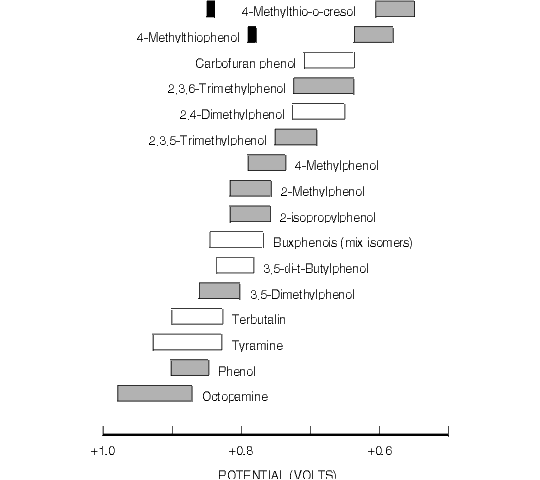
Figure 8.4. Oxidative Cyclic Voltammetric Data for Selected Chlorophenols of Environmental Interest.
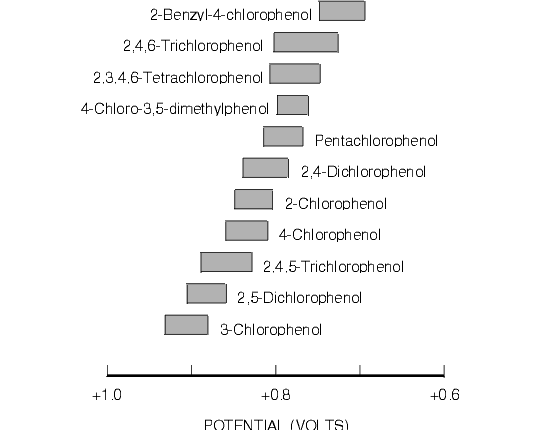
Figure 8.5. Oxidative Cyclic Voltammetric Data for Some Vanillyl Metabolites of Tyrosine and Related Compounds.
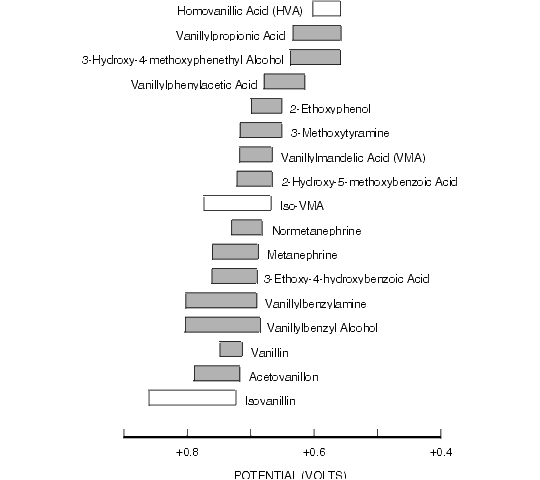
Figure 8.6.Oxidative Cyclic Voltammetric Data for Some Indole Metabolites of Tryptophan and Related Compounds.
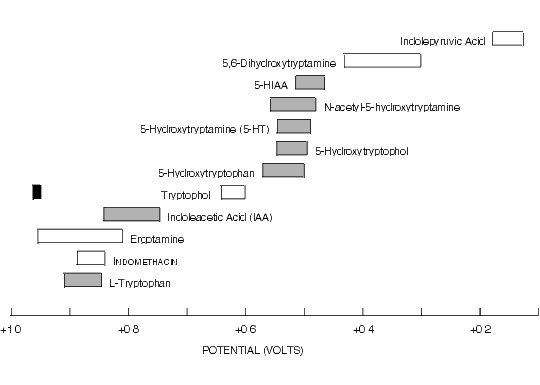
Figure 8.7. Oxidative Cyclic Voltammetric Data for Biologically Important Catecholamines and Other Easily Oxidized Diphenols.
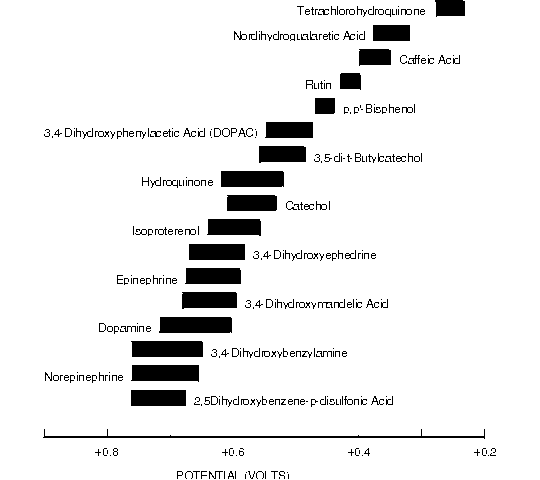
Figure 8.8. Oxidative Cyclic Voltammetric Data for Some Natural Phenolic Acids.
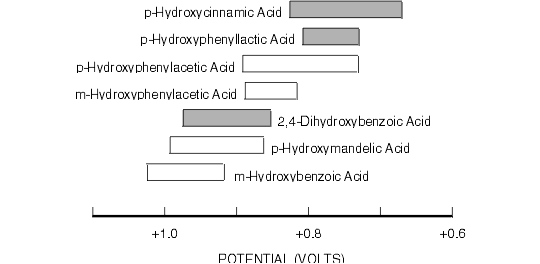
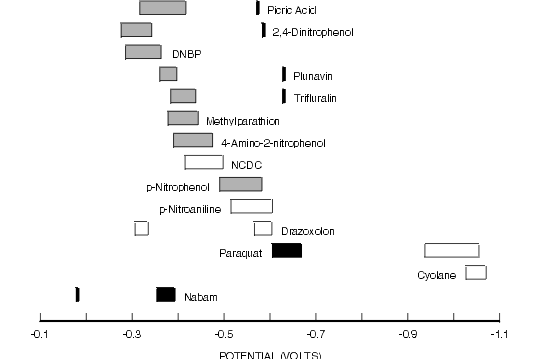
Figure 8.9. Reductive Cyclic Voltammetric Data for Selected Compounds of Environmental Interest.